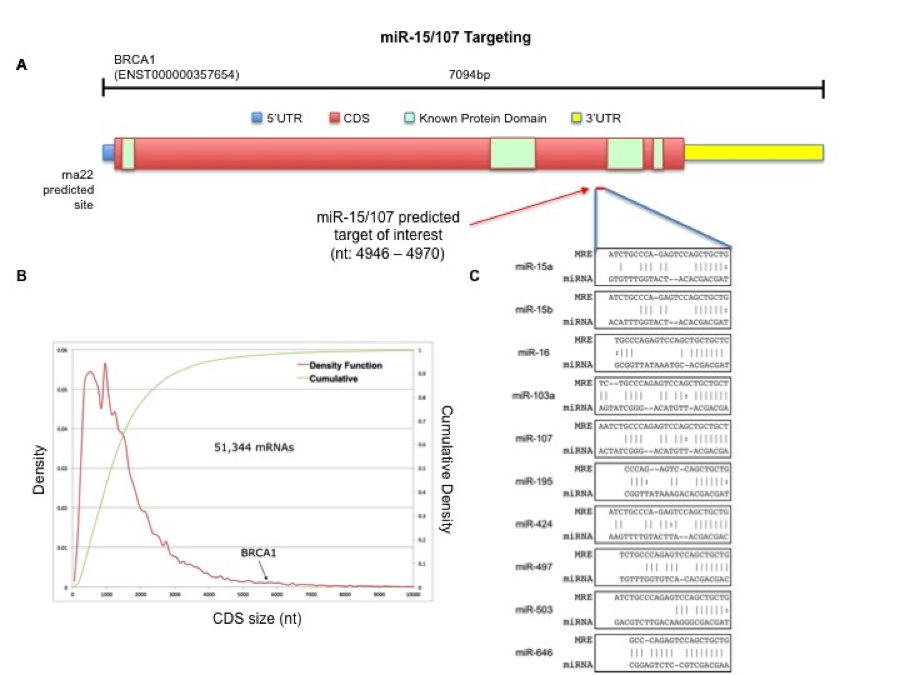
BRCA1, a major tumor suppressor gene responsible for coordinating the repair of double-stranded DNA breaks and maintaining overall genomic stability is commonly dysregulated in many cancers, a process which up until recently, has largely been attributed to loss-of-function mutations resulting in inactivated BRCA1 protein. Interestingly, at over 5.5 kilobases, the BRCA1 coding sequence is among the longest 2% of all protein-coding genes in the human genome, and while there have been reports of post-transcriptional regulation of this gene through standard miRNA interactions within its 3′-untranslated region, there has not existed any evidence for non-standard miRNA regulation taking place within the BRCA1 coding sequence, despite its substantial length.
The miR-15/107 group, which consists of 10 miRNAs that share a common sequence at their 5′ seed-region, are evolutionarily conserved across vertebrate animals and found in increasing numbers among mammals, with primate and humans genomes being the only known to harbor all 10 miR-15/107 members. Importantly, miRNAs from this group are collectively expressed ubiquitously across tissue types, are known to regulate many cellular processes such as cell proliferation and metabolism and have been demonstrated to participate in non-standard interactions outside of the 3′-untranslated region of target mRNAs.
Using the miRNA target prediction tool rna22, we uncovered a putative target site for the miR-15/107 group of miRNAs within the coding sequence of BRCA1. By over-expressing the miR-15/107 group of miRNAs in a panel of 10 cell lines representative of both normal and cancerous breast, colorectal and pancreatic tissues, we observed durable suppression of BRCA1 transcript levels. Furthermore, we confirmed the specific interaction of these miRNAs upon the BRCA1 coding sequence by co-transfecting miR-15/107 precursors with a luciferase expression plasmid containing the putative target sequence. Interestingly, when we tested the effects of these miRNAs on BRCA1 protein levels in a smaller subset of cell lines, we found variable suppression of BRCA1 protein in a tissue-specific manner, suggesting a significant degree of cross-talk exists between the miR-15/107 group and mediators of BRCA1 transcription, translation, and protein-stabilization. Together, these findings highlight the degree to which non-standard miRNA interactions may influence DNA damage responses differentially across tissues and could potentially have implications for recent therapeutic strategies aimed at exploiting defective DNA damage response pathways in cancer.
 miR-15/107 family targeting of BRCA1
miR-15/107 family targeting of BRCA1
References
- Quann K, Jing Y and Rigoutsos I (2015). Post-transcriptional regulation of BRCA1 through its coding sequence by the miR-15/107 group of miRNAs. Front. Genet. 6:242. doi: 10.3389/fgene.2015.00242
